A Fever in the Dust
Arthur Charles used to start each day with a morning walk with his wife. The loop around his neighborhood in Bakersfield, California wasn’t usually difficult for the 50-year-old to complete. Then one day, two years ago, Charles could barely reach the corner of his street. The routine walk felt like he was running a marathon.
“I couldn’t breathe. I was tired,” he recalls.

Arthur Charles is a resident of Bakersfield, in the southern Central Valley of California. He works as a major league baseball recruiter and rec specialist at West Side Recreation and Park District. Credit: Kerry Klein/KVPR
Charles visited his doctor, who thought he might have pneumonia, and prescribed a round of antibiotics. Charles was sent home. Later that evening, he was pouring sweat, running a fever, and vomiting. Unexpected trips to urgent care continued for three weeks—but no one could figure out what was wrong. One evening, his wife noticed that he was barely breathing while he dozed off. They rushed to the hospital, where Charles' oxygen levels were alarmingly low. “They said if I would have went to sleep that night, I wouldn't have woken up.”
After he was admitted to the hospital, his symptoms—shortness of breath, vomiting, dizziness, and extreme exhaustion—grew worse.
"That was the first time I’ve ever felt like I can’t do anything. I just felt helpless," he says. "I've never felt so weak and so beat in my life."
Finally, after several misdiagnoses, a blood test revealed that Charles did not have a bacterial or viral infection. He had breathed in a fungus that had hijacked his lungs—and was headed for the rest of his body.
The doctor told Charles’ family he might die.
DEATH BY FUNGUS
Charles had Valley Fever, which he continues to battle today. This disease, medically known as coccidioidomycosis, is caused by the fungus Coccidioides, or Cocci for short. The dangerous fungus lingers in the soil and dust in the American West, as well as Mexico, and Central and South America. Anyone who lives, or even passes through, these regions has a chance of becoming infected.
Valley Fever isn’t contagious. It doesn’t spread from person to person like the common cold or flu. All it takes to get Valley Fever is a single breath of the fungus when it’s swept up by the wind.
The latest numbers show that in 2018, about 15,600 people were diagnosed with Valley Fever in the United States, with most cases coming out of southern Arizona and California’s San Joaquin Valley. But these numbers only reflect known cases reported to the Centers of Disease Control. “That number is probably a drastic underestimate of the true number, because people and doctors are not looking for this disease,” says Orion McCotter, a former CDC epidemiologist who tracked outbreaks of Valley Fever.
Even though Valley Fever isn’t well-known in other parts of the country, if you live in endemic regions, it’s likely that you know someone who’s had the disease. I grew up just outside of Fresno, and have family living all through the Central Valley, an agriculture cradle surrounded by a fortress of periwinkle mountains. Many of my relatives have worked the fields and orchards that sprawl between the valley’s quickly-growing cities.
My mother grew up in the small town of Dinuba, and she contracted Valley Fever when she was a teenager. She was working her uncle’s onion farm in Madera one summer, unloading bin after bin of the dried red bulbs. “Every time you dumped one out, you could see all the dust flying in front of you. Every day we’d be inhaling it, inhaling it,” she tells me.
She started having coughing fits at night. She brushed it off as a cold at first, but then her lungs began to rattle and her fever began to climb. She noticed strange red-purple lumps on her legs that she confused for bug bites, and that wouldn’t go away. Then she became unusually tired.
“I thought, this is not my norm. I didn't know what it was.”
The most common symptom of Valley Fever is exhaustion. Many people who get infected have symptoms similar to the flu—a mild cough, a low fever. Usually, these people can fight off the infection on their own. They may not even realize they were sick with the fungus.
“A lot of times, when people hear about fungal diseases, they think about toenail fungus and skin infections,” says McCotter, now an epidemiologist for the state of Oregon. “But really, fungal diseases can cause very severe infections.”
Approximately 40% of Valley Fever patients develop a pneumonia-like condition that requires medical attention. My mother was able to fully recover after doctors identified she had Valley Fever, but others, like Charles, may get treated for the wrong disease. Valley Fever is a commonly misdiagnosed illness because many symptoms are similar to the flu and pneumonia—an issue we've also been seeing with COVID-19. It can even be mistaken for lung cancer, as the fungus can create nodules and scarring in the tissue, which look similar to tumors in a chest X-ray.
But in some severe cases, the fungus can leave the lungs and spread to other parts of the body. It can invade the skin, bones, nervous system, and even the brain—turning into disseminated disease. Without proper antifungal treatment, the disease can last a lifetime, or even be deadly.
For these patients, “it’s either a death sentence, or a life sentence,” says Anita Sil, a microbiology and immunology professor studying fungal diseases including Valley Fever at the University of California, San Francisco. “Their lives are changed forever, and they’re on antifungals forever. They don’t get their normal health back, so the consequences are huge.”
People with severe Valley Fever can lose their ability to work. Some lose limbs. Others lose their homes to pay medical bills. And in the most dire cases, what’s lost are their lives. These severe, disseminated cases of Valley Fever are rare, only about 1%. But, it is more common among immunocompromised patients, women in late stage pregnancy, and African Americans, Filipinos, and Native Americans, according to the CDC and public health departments.

The cobweb-like shadows in this chest X-ray are signs of pulmonary fibrosis from Valley Fever. Since these shadows also resemble those seen in other lung diseases, including tuberculosis or lung cancer, a chest X-ray needs to be coupled with other testing, as well as possible tissue biopsy. The amount of scarring found in the X-ray can show the severity of the fungal infection. Credit: CDC/Public Domain
The many ways the fungus can make people sick has long puzzled scientists studying the disease. Why does the fungus cause a cough in some people, but a deadly meningitis in others?
“I've always been interested in immune dysfunction,” says Katrina Hoyer, an immunologist and assistant professor at the University of California, Merced. “What is different about the immune response in the people that get severe disease, and what is wrong with that immune response? Can we manipulate it?”
Researchers like Hoyer are racing to understand the fungus better—especially as the pathogen is moving north and east. They’re trying to discover how the fungus grows in the soil to better understand where Valley Fever may expand in the future as the climate warms.

A scanning electron microscope image of the spherules of Coccidioides posadasii, one of the species of fungus that causes Valley Fever. The species is named after Argentinian physician Alejandro Posadas who was the first to identify the disease in a 33-year-old cavalryman in San Juan in 1892. Credit: Bridget Barker
THE IMMUNE SYSTEM WAGES WAR
Coccidioides is a shapeshifter: It has one form in the soil and another in the host’s lungs.
Many fungi can change their structure under certain conditions, an ability known as dimorphism, explains Anita Sil at UCSF. Cocci belongs to a group of fungal pathogens called thermal dimorphs, which change their cell shape in response to temperature. Once the fungus is inhaled, temperature differences between the environment and the mammalian host help trigger Cocci’s transformation, Sil says.
“When they get into a mammal, they change their program entirely, the shape of the cells changes dramatically,” Sil says. It morphs into a large capsule, called a spherule, that is filled with spores. These changes mean the fungus is ready to invade.
The body’s immune system wages war. Normally, a variety of immune cells each have different jobs. Cells on the defensive line are watchdogs for pathogens and signal the alarm when they enter the body. Some immune cells are like generals, in charge of directing and turning on immune responses. Others are like soldiers, battling against specific intruders. The body’s immune cells typically would engulf a bacterium or a virus to fight the infection. But these fungal spherules are big, making them difficult to swallow.
“It’s sort of like a King Kong-type mechanism, like you have this enormous beast that is too big for mere mortals to conquer,” Sil says.
If not stopped, the spherules can pop like an overfilled balloon, unleashing more of the fungus to migrate through the body.
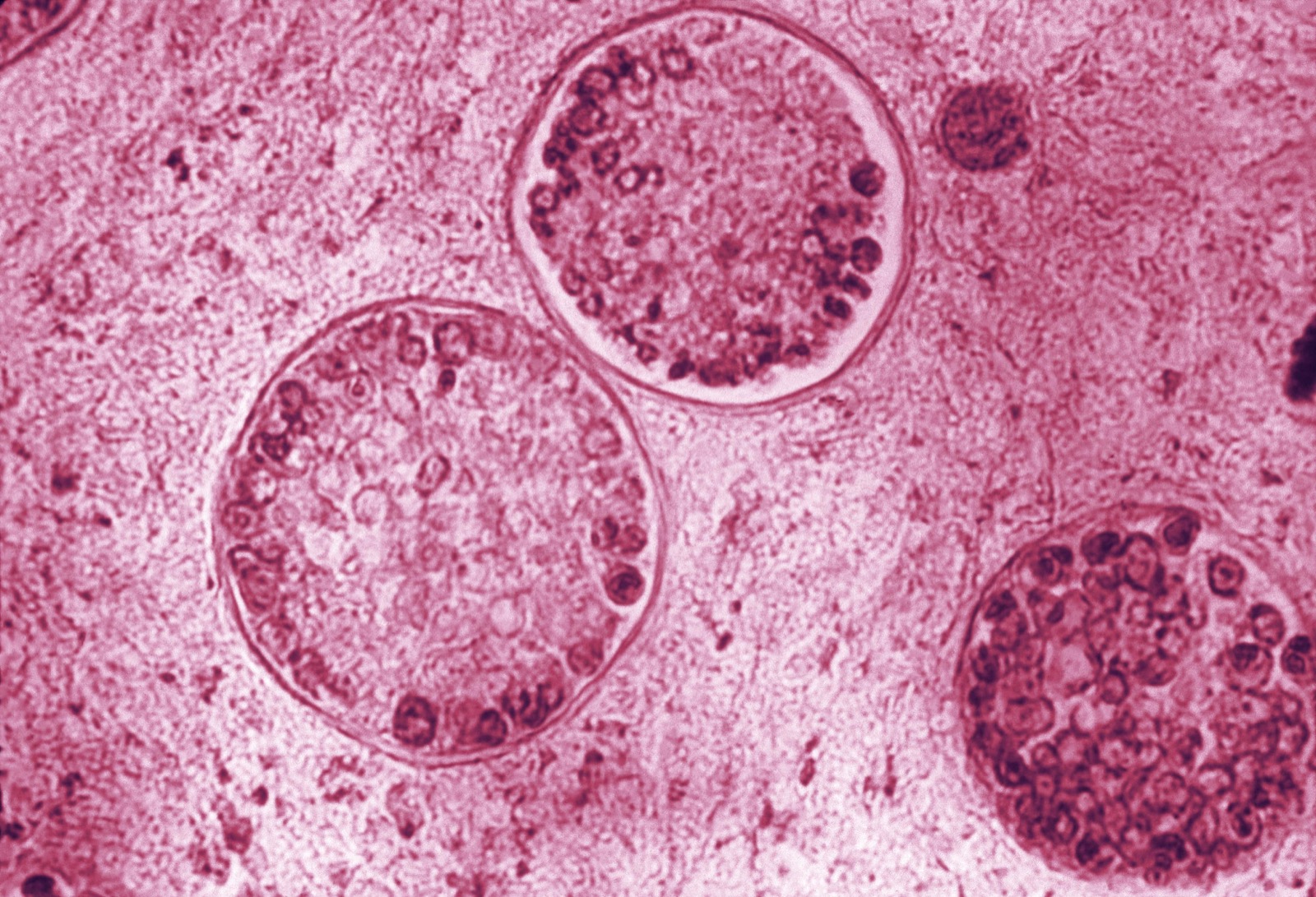
This photomicrograph reveals the spherules of one of the species of Valley Fever fungus, Coccidioides immitis. Within these spherules, you can see the spores. Credit: CDC/Public Domain
Antifungal medication is the only treatment doctors know can quell Valley Fever. It works best if the disease is caught early. Years before Charles got sick, his middle son had Valley Fever in 2008. His doctor quickly diagnosed and treated him, tipped off by the strange lesions growing on his legs—a symptom that Charles himself did not develop. His son made a full recovery after a year of antifungal medication.
But some people say that the antifungals are as bad as the disease itself, if not worse. Since the biology of fungi is very similar to the biology of our cells, antifungal medication can also cause damage while attacking the invader, says Katrina Hoyer at UC Merced. It leads to a cache of brutal side effects—constant dry skin, nausea, abdominal pain. Some patients with severe Valley Fever receive an IV of the antifungal amphotericin B, nicknamed “amphoterrible” for causing side effects akin to chemotherapy.
The other problem with antifungal drugs is that they don’t always destroy the fungus. They only inhibit the growth of the fungus enough for your immune system to try to kill it.
Despite treatment, Charles' immune system can’t eradicate the fungus completely. He must continue to take antifungal medication to control its growth. He is prescribed one of the more common antifungal drugs, fluconazole (Diflucan). He points to a swollen crack on his lower lip, a result of constant dry skin and dehydration from the medication.
“I think I should have stock in chapstick because I use so much of it,” Charles says. “There's just so many different side effects to the medication that it's ridiculous. But I have to take it if I want to live.”
In the worst cases, the fungus proves near impossible to control. This is what happened with Charles’ older sister, Deborah.
“My sister was amazing. She was athletic, she loved to cook and bake,” Charles says. He likes to brag that he was Deborah’s favorite sibling. “She would bake all my favorite desserts. She would make a thing called a sock-it-to-me cake, pineapple-upside-down cake, pound cake.”
But a few years after graduating college, Deborah was hospitalized. She was misdiagnosed and given antibiotics multiple times before doctors concluded she had Valley Fever.
“When they figured it out, it was too late,” Charles says.
The fungus had already wreaked havoc in her body. The spores proliferated, spreading from Deborah’s lungs, to her bones, to her spinal column, to her brain. It caused her to develop meningitis, a severe swelling of the brain’s membrane. Deborah lost her eyesight. Her weight dropped from around 165 pounds to about 70 pounds.
“She lost everything,” Charles says. “To watch her go from being vibrant and beautiful and strong as ever, to almost being an infant again was tough.”
Deborah was only in her mid-twenties when she died from Valley Fever.
Charles’ family has been hit three times by Valley Fever. Each case shows how different the progression of the disease can be.
THE MYSTERY OF MISFIT IMMUNE CELLS
In her lab, Hoyer is investigating differences in the immune response of Valley Fever patients—watching for red flags when it goes awry. She surveys anomalies in the numbers and behaviors of immune cells. In a 2018 study with pediatricians at Valley Children’s Hospital in Madera, Hoyer was able to narrow in on a specific type of immune cell: regulatory T cells. “Their job is to be the brakes on the immune system,” says Hoyer.
She and her colleagues took blood samples from 20 children who were healthy, and 30 children who were infected with Valley Fever, and found a potential key difference in their cell numbers. “It looks like patients that will go on to have chronic or more persistent infection have a higher frequency of these regulatory cells.”

At UC Merced, Katrina Hoyer (pictured first), graduate student Anh Diep (pictured second with Hoyer), and colleagues harvest avirulent strains of the fungus. They're doing the fundamental research needed to understand the immune response in severe Valley Fever cases. Credit: Lauren J. Young
More regulatory T cells could mean hitting the brakes on the immune response needed to defend the body, allowing the fungus to run amok. But Hoyer and her team are still trying to find out what could be causing the higher levels of regulatory T cells in severe cases.
“These patients may either have more regulatory cells to begin with, or they are inappropriately developing or expanding their regulatory [cells] instead of the cells that are needed to fight the infection.”
Other labs have also looked at misbehaving T cells that fight off pathogens. For example, in 2018, clinicians at UCLA were treating a four-year-old boy with severe Valley Fever when they noticed his T cells were acting strangely. At that point, he was very sick with a high fever and large skin lesions on his back and skull. The boy’s T cell counts were normal, but they appeared to be fighting the wrong pathogen.
“If you’re fighting werewolves, you need silver bullets. Everybody knows that,” says Manish Butte, the pediatrician and UCLA professor who treated the young boy. “If you hit them with regular steel bullets, that’s not going to work.”
For certain threats, you need specialized defenses. But if you’re given the wrong message of what you’re up against, your attack will be ineffective. In this case, instead of gearing up to fight a fungal invader, the boy’s T cells were armed for a battle with an allergy or a parasite, such as a worm or a tick. The cells, Butte says, had “totally the wrong idea.”

Six-year-old Abraham Gonzalez-Martinez at a checkup at UCLA in November 2019 along with clinicians Maria Garcia-Lloret (center) and Manish Butte (left). Read and listen to Kerry Klein's feature story following Abraham's treatment on KVPR Valley Public Radio. Credit: Nick Carranza/UCLA Health
Butte and his team were able to find a misstep in the messaging to the T cells: a lack of a signal protein called Interferon gamma in the boy’s body. The protein is used to initiate the type of immune response needed to fight the fungus. At first, the doctors gave the boy a boost of the protein. It helped kickstart the right immune response, but the wrong attack response was still running, Butte says. So next, the team turned to a drug typically used to treat eczema that blocks that immune response. By giving the boy both the signal protein and the drug, the doctors were able to steer the cells towards the right immune response—send the correct message to load the right bullets.
Over the course of about six weeks—the length of time it typically takes for these types of therapies to kick in—the boy’s skin lesions melted away, his inflammation calmed down, and he began to recover.
“His cells were just responding,” says Butte. “It was a crazy punch that his immune system needed to suddenly say, ‘Oh my God, we have to fight this infection’ and it did in just a gangbuster sense.”
The team’s work is a step towards a new, effective treatment. For now, it’s only been tried in one patient. Butte and his team at UCLA are working with Kern Medical in Bakersfield to roll out a large clinical trial. Until then, Valley Fever patients still have to wait for a proven cure.
But the mysteries actually start long before patients need treatment. Coccidioides survives through a journey before it makes a home in a patient’s lungs. What if scientists could intervene well before the war waged by your immune system begins—back even before patients are exposed?
Anh Diep studies Coccidioides in the lab. Read more about the surge in Valley Fever research at California universities on KVPR Valley Public Radio. Credit: Lauren J. Young
A TERROR IN THE BODY, A "WIMP" IN THE SOIL
The life of the Valley Fever fungus, and the root of the problem, begins beneath our feet. There are currently two known species of Coccidioides, and both cause disease: Coccidioides immitis, is found in California and in Washington state, and Coccidioides posadasii, which is found all over the western part of North America and parts of Central and South America. Both Cocci species tend to grow in the top layers of the earth, and like alkaline, salty soil—like the hard packed clay and sandy loams that makes up much of the American Southwest.
“The [Valley Fever] hot spots are in what we call the former Bakersfield deserts in the southwest,” says Antje Lauer, a microbial ecologist at California State University, Bakersfield.
Little organic matter typically exists in this soil. It’s a hostile environment for many microorganisms to live in, but this is where Coccidioides thrives. The fungus could grow in more organically rich soils if given the chance, says Lauer, but these environments are dominated by other microbes that often outcompete Cocci.
“It's a little wimp,” Lauer says, and adds, “That's often the case with a lot of dangerous pathogens.”
Cocci may become a bursting bulbous sphere in the human body, but it looks drastically different in the soil. During a rainy winter or monsoon season, the fungus will begin to grow in long, filamentous strands, called mycelia.
“I think it looks like fuzzy brie,” says Anh Diep, a research graduate student in Hoyer’s lab at UC Merced, where the team keeps a mini-fridge-sized incubator filled with the fungus. Stacks of petri dishes are completely covered in puffy, cottony domes—like the fluffy stuffing that you’d pull out of a plush toy.
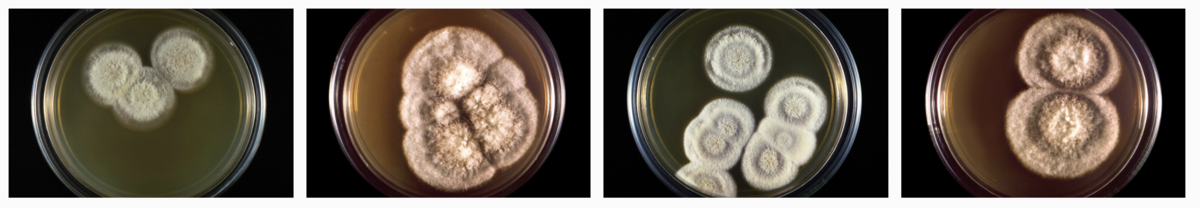
Plates of Coccidioides immitis. Credit: CDC/Dr. Libero Ajello/Public Domain
Although researchers have been studying how Coccidioides grows in its environment since the 1930s, scientists still don’t understand why it seems to like particular patches of dirt. “I can literally go to [one] spot year after year after year, and always find the fungus there, but I go two meters away and I don’t find it,” says Bridget Barker, a fungal ecologist and associate professor at Northern Arizona University in Flagstaff. “What is it that’s driving that really disjunct distribution in the environment?”
In desert regions, Barker pays attention to the plants and animals—any common factors that could influence the fungus. Recently, she found one. Barker and her team noticed that they kept getting positive tests for Coccidioides in soil samples taken near burrows of desert rodents, like pocket mice and kangaroo rats. These burrows might be creating perfect homes for the fungus, Barker says.
“These nice, little microenvironments have higher humidity, lower temperature so the fungus is maybe a little happier,” she says, and “the hair, the skin, the feces, the urine—whatever they’re leaving behind in the burrow environments is a good food source.”
The rodents could also potentially serve as a carrier for the disease—getting infected, dying, and decaying in the soil provides more nutrients for the fungus to feed on, she says. Now, she is teaming up with wildlife biologists at Arizona’s Game and Fish Department to collect blood samples from rodents to see if they are infected with or have been exposed to Valley Fever.

Barker (right) and her team sample in the field. Credit: Bridget Barker
Barker is not the first to make this observation. Researchers at UC Berkeley have also found similar associations in California. Finding out what is driving the growth of the fungus in these burrows could inform people’s decisions about what plots of soil to leave alone. This could help lower the chances of releasing the fungus into the air.
“If we can understand certain times of the year, or certain areas that people are going to be more at risk for exposure than I think we can also tackle the disease in that way as well,” Barker says.

The setting sun highlights a dusty haze that lingers in the San Joaquin Valley. Taken in Fresno on November 25, 2019. Credit: Lauren J. Young
THE DEMOGRAPHIC DANGERS OF DUST
The mountain ranges of the Central Valley often cup a mustard haze. At dusk on a bad air day, you can see a brown soup of smog, pollution, fine particles, and dust. One day in November, while I was visiting family in Fresno, I got caught in the middle of strong winds. The dust clung to my clothing and hair. I could even taste and feel the gritty grains on my teeth. A “severe alert” notification buzzed on my phone’s weather app: “The Valley Air District has issued an Air Quality Alert… due to blowing dust caused by windy conditions.”
“Severe Alert” in Fresno on November 25, 2019. Credit: Lauren J. Young
These alerts aren’t uncommon. Canceled recess and rescheduled sports practice due to poor air quality and dust storms were the norm in my childhood. The conditions are a trigger for asthma—and prime for Valley Fever. On these particular days, Charles and his children stay indoors. But if he must go outside, he’ll put on a protective garb that many more people are wearing out today: face masks.
“We used to laugh at people who ride around in cars with the mask on or walk down the street, like ‘why do you got the mask on?’ But now we're those people,” says Charles. “It’s just something as simple as just a little surgical mask, but cover yourself up and try to prevent from breathing in the spores, because it doesn't take much.”
The mycelia of the fungus dry out, and the thin walls of their spores called arthroconidia easily break. These spores get carried in the air with disturbed dust, which scientists call “fugitive dust.” Any disturbance to the earth will do. Natural causes like wind storms and even earthquakes have been linked to cases of Valley Fever. In 1977, an outbreak of 379 new cases followed a significant dust storm that blew through Kern County, while in 1994, Ventura County experienced a caseload 10 times its average after the Northridge earthquake stirred Cocci into the air.
Increasingly there are also unnatural, human disruptions: construction of new housing, schools, buildings, solar panel fields, highways. Unpredictable, frequent bouts of fugitive dust is a growing problem in developing cities in the American Southwest, like Kern County’s Bakersfield.
“People look at Bakersfield, and they think it’s just a hole in the wall, like it’s just not much when you come from the big city,” says Charles. But “we’re growing.”

A dust storm hits in Arizona. Credit: Ms. Phoenix/Flickr/CC BY 2.0
The expanding southern Central Valley city is flanked by farms, an industry commonly linked with Valley Fever. The very first cases documented were in agriculture workers and immigrant farmers in the 1800s. However, farmers are often wrongly blamed for the spread of the Valley Fever.
“Many people believe that the fungus is spread because of agricultural practices. In reality, the fungus is found in soil that is uncultivated,” says Hoyer. Agriculture fields rich in organic matter, such as plants and fertilizer, help promote the microbial diversity that outcompetes Coccidioides. However, when farms are abandoned and lie fallow, it provides a chance for the fungus to return.
“We have this a lot now in the Central Valley, that farmers are not able to farm anymore because they can't afford the water, especially the small farmers,” says Lauer. “The spores might actually germinate, and then we have a Valley Fever site again.” She had found this cycle happening in one of her soil testing sites at a former farm in the Lancaster area, near Bakersfield, which turned out positive for the fungus, she says.
Outdoor workers on farms and construction sites do have a higher risk of exposure, being out next to nearby abandoned fields or constantly breathing in clouds of dust from a development project.


Fresno, California. Credit: Lauren J. Young Bakersfield, California. Credit: Lauren J. Young
“The Central Valley is this agricultural epicenter, but also this developmental epicenter,” says Diep. “So when we think about the demographics that are involved most heavily in these occupations, they tend to be Latino or Hispanic immigrants and minorities.”
On top of a socioeconomic barrier and cultural divide, Diep says, poor access to healthcare can delay proper treatment for Valley Fever, allowing the disease to become more severe: “If we think about all the counties in California, the San Joaquin Valley has one of the lowest numbers of primary care physicians and specialty care physicians per 100,000 people in the state. And so we already have a lack of these clinicians and physicians who could see patients.”
Living in an endemic hot spot and working outside isn’t the only danger. Some groups also have an elevated risk of developing the more severe, disseminated version of Valley Fever: certain racial minorities, including Filipinos, Native Americans, and African Americans—like Charles, his son, and his sister. It’s a disparity that researchers say is not simply caused by higher exposure.
“Anyone who is exposed to the fungus has the same likelihood of getting infected, but some populations are more likely to develop the more severe disseminated disease,” says Hoyer.
Hoyer, Butte, and other researchers suspect it could be a difference in the genes that code for the immune cells. There could be something in the immune genes that causes the body to be less effective at mounting the right type of immune response to fight the fungus, Hoyer says.
“Skin color, obviously, is unrelated to infection,” says Butte. “It's the immune genes that we want to understand. And if some of these [historical reports that] African Americans are more infected is false or true, we need to understand why. The genetics are going to be really key.”
A GROWING THREAT WITH CLIMATE CHANGE
Better understanding the disease will be essential, as Valley Fever numbers have been climbing, and show no signs of stopping.
“Our numbers are on the rise. The past three years, we've had record years for infections,” says Hoyer.
California documented over 7,500 cases of Valley Fever in 2018, the highest of all states. In 2018, in Kern County, Valley Fever numbers rose for the fourth year in a row, according to the most recent data. Increasing rates have been seen in Arizona, Nevada, New Mexico, and Utah.
These southwest states have some of the country’s fastest growing cities. As populations increase, so do the number of cases. Health clinicians may also be getting better at reporting and diagnosing the disease—some states only began keeping records of Valley Fever in 1992.
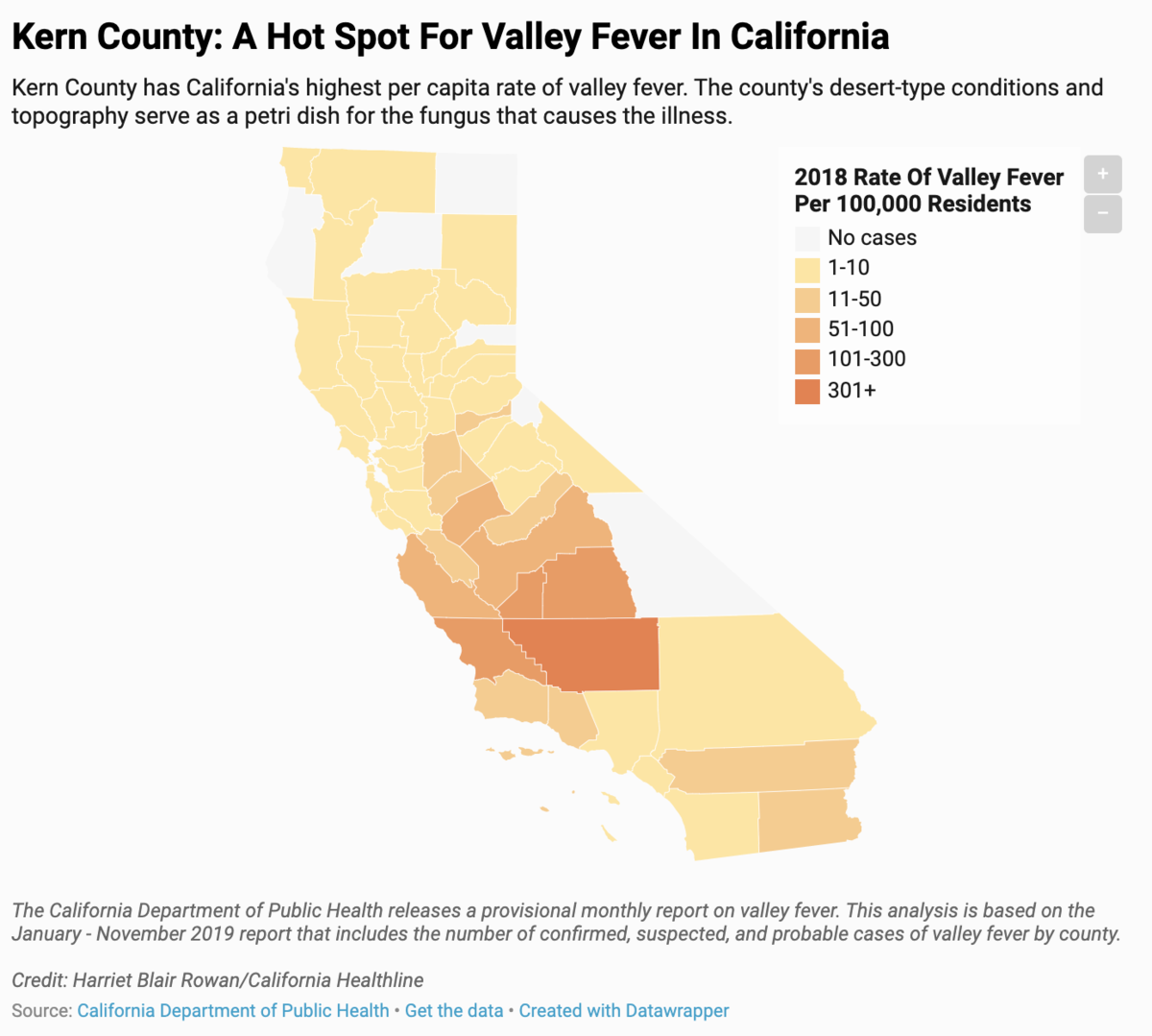
Credit: Kaiser Health News/CC BY-NC-ND 4.0
But the fungus itself also seems to be spreading. As hot, arid environments expand with climate change, Valley Fever territory could, too.
Valley Fever was reported in Washington state for the first time in 2014. Soil tests came back positive for the fungus, confirming that these cases originated from within the northwest state. It surprised many researchers that the disease could be found so far north. It may soon move even further, as the climate becomes more favorable for the fungus across the United States.
“Valley Fever is an example of a disease that will probably be more widespread from climate change,” says Morgan Gorris, a postdoctoral research fellow at Los Alamos National Laboratory and a former graduate student at UC Irvine, who maps out environmentally influenced diseases, like Valley Fever. “I anticipate we will see larger health impacts from this disease in the future.”
Current maps of the disease are incomplete at best. Valley Fever is not required to be reported to the CDC. Texas, a state on the edge of the endemic region, chooses not to report Valley Fever cases nationally.
“Texas has been known to have cases since the 1930s,” says Gorris. “Some counties do report their case counts, but the whole state does not. It's not mandated. I feel like I would have a lot better understanding of where the boundary of this disease is if I had case data for Texas.”
The CDC’s maps are based on skin test data, a test which revealed if you had been exposed to the fungus. While useful, that data was published in the 1950s, with a handful of updates from available outbreak numbers and studies. “The population makeup was very different then,” says Hoyer. “We also don’t know exactly the endemic region anymore.”
Read more about the study at KVPR Valley Public Radio. Credit: Morgan Gorris/UC Irvine/GeoHealth
Gorris and scientists at UC Irvine created a new map to show where Valley Fever might expand by 2095, published in the journal GeoHealth in August 2019. The map used current and future climate factors, including both temperature and rainfall. Places likely to become hot and dry, like the endemic areas today, show up on the map in hot pink. That hot pink reaches as far north and east as North Dakota—well within the midwest. Under the highest climate warming scenario, Valley Fever’s endemic region could more than double and the number of cases could increase by 50% by 2095.
“In the U.S., I think Valley Fever will be high up on the importance list, especially if the endemic region expands the way our model projects it will,” Gorris says.
While it may look ominous, this map can help healthcare workers prepare. Public health officials can create better surveillance and disease awareness now that they can anticipate which counties and states will begin to develop cases where they may not have before, Gorris says.
“Your time to diagnosis can be decreased, and your chances of a more serious form of the disease decrease,” she says. “So, really, although it is projected to possibly impact more people, we want this to be used as a tool for preventing those infections, or at least mitigating the health impacts.”
Coccidioides isn’t the only fungal disease that may benefit from climate change, spreading beyond its original home. Some scientists suggest that global warming could cause fungal pathogens to become a greater threat to humans. There are over five million species of fungi. While only a few hundred of those cause disease, fungal pathogens are responsible for approximately 1.6 million deaths every year.
“There's a lot of attention that is placed on viral diseases and bacterial diseases, but fungi are everywhere,” says McCotter, who has witnessed Valley Fever first hand in his dad and pet dogs growing up in Tucson. “I think that [fungal pathogens] are definitely under recognized.”
The warmer temperatures might also change the way the fungus works in our bodies. One of the first lines of defense against the fungus is our body temperature. Our internal temperature helps us kill off pathogens. It’s why we get fevers when we get sick; it’s the body’s way to prevent the rapid growth and spread of the pathogen, explains Hoyer.
“That poses a challenge for the fungus, and gives our immune system a fighting chance,” says Hoyer.
If the fungus adapts to the warmer temperatures, it’ll make it tougher for our bodies to kill the fungus. “That reduction in that difference in temperature [between the environment and our bodies] is going to be more of a problem,” she says.
Valley Fever might soon be affecting many more of us. But research scientists and survivors like Charles are keeping a close eye on its movement, spreading awareness to help mitigate the impact of the disease.
“It's not a disease that is restricted only to the southwest,” says McCotter. “It's something that's important for clinicians and the public to be aware of across the U.S.”
"IT WON'T LET GO"
Arthur Charles is out on the frontlines helping others in his community understand the severity of the disease. In February in Taft, a city 45 minutes from Bakersfield, Valley Fever survivors and supporters stretch and warm up for a 5K run and 2K walk. Kern Medical’s Valley Fever Institute and the West Side Recreation and Park District put on the event to help spread awareness of the disease—and it was all Charles’ idea.
“I wanted to have it out here because it affects so many people,” he says.

Arthur Charles helped host the first annual Valley Fever 5K Run and 2K Walk, with Valley Fever Institute at Kern Medical. Listen to participants and learn more about the event in an audio story on KVPR Valley Public Radio. Credit: Kerry Klein/KVPR
Charles has lived nearly his whole life in Bakersfield. Growing up, he played baseball and was on his high school’s first basketball team to win the state championship. He runs around Kern County, recruiting baseball players as an associate scout for the Tampa Bay Rays, and works as a rec specialist for West Side Recreation and Park District.
But today, his Valley Fever can leave him breathless and zapped of energy. Some days, it’s hard to walk to his mailbox at the end of his street, let alone make it to work. “I always would stay busy and do activities, and now, I just take it one day at a time and see what I can do,” he says.
For now, Charles continues to take his antifungal medication. Until there’s a cure, he’ll likely need them for the rest of his life. “It won't let go of me,” he says.
But he isn’t giving up.
"I'm a fighter. So this is gonna be a battle, you know? If I go out, I'm gonna go out swinging and that's for sure.”



